Q-Line Biotech Ltd. (QBL) is engaged in the business of developing, manufacturing and marketing of diverse range of reagents (including kits and POC devices) & consumables and manufacturing, importing, distribution/supply of diagnostic equipment for different diagnostic healthcare needs. The company supplies diagnostic equipment and IVD products for different diagnostic healthcare needs since 2013 directly or through its distributor/s majorly to diagnostic service providers, hospitals and medical colleges.
The company has established its brands over a period of 12 years through its experience, R & D, manufacturing capabilities and quality assurance. The core segments of operations of the Company in IVD Industry include Clinical Chemistry, Haematology, Immunodiagnostics, Molecular Diagnostics and Others (POC Devices & Rapids).
QBL’s key manufacturing segments include indigenous manufacturing of reagents including Clinical Chemistry, Haematology, Immunodiagnostics, Molecular Diagnostics and Others (POC Devices & Rapids) and supplying/ manufacturing of in-vitro diagnostics (IVD), Pathology equipment’s & devices. Further during the Covid-19 pandemic, the company diversified its focus and with the technical collaboration of third-party institutes and through its own R&D team developed a range of Covid testing kits viz. RT-PCR Kits, RNA Extraction Kits, VTM Kits etc.
It is research driven company engaged in developing and manufacturing a wide range of reagents formulations used across various IVD and diagnostic needs. The company leverages its R&D capabilities to develop and manufacture a portfolio of differentiated reagent formulations /products. Further, for its certain Class of Reagent & equipment’s and devices manufacturing business, the company has entered into technical collaboration with certain international companies. Under the agreement terms, it undertakes the manufacturing of these Reagent and equipment’s and devices as per the technical collaboration and specifications provided by the partners or companies.
With the help of these collaborations the equipment and devices adhere to strict quality control, international standards and certifications. As of March 31, 2026, the company employed 19 personnel at R&D laboratories, which constituted 5.25% of its total permanent employee strength. As of March 31, 2026, it had 362 employees on its payroll and additional 223 contract employees in various departments.

The company is coming out with its maiden book building route IPO of 6253200 equity shares of Rs. 10 each to mobilize Rs. 214.48 cr. at the upper cap. The company has announced a price band of Rs. 326 - Rs. 343 per share. The minimum application to be made is for 800 shares and in multiples of 400 shares thereon, thereafter. The IPO opens for subscription on May 21, 2026, and will close on May 25, 2026. The IPO constitute 26.81% of the post-IPO paid-up capital of the company. The shares will be listed on NSE SME Emerge. From the net proceeds of the IPO, it will utilize Rs. 93.50 cr. for working capital, Rs. 90.00 cr. for repayment/prepayment of certain borrowings, and the rest for general corporate purposes.
The company raised Rs. 27.44 cr. in a pre-IPO placement of 800000 shares in May 2026, at Rs. 343 per share.
The IPO is jointly lead managed by Hem Securities Ltd., and Share India Capital Services Pvt. Ltd., Purva Sharegistry (India) Pvt. Ltd., is the registrar to the issue. HEM group’s Hem Finlease Pvt. Ltd., is the market maker as well as a syndicate member.
The company has issued initial equity capital at par value. It raised further equity shares in the price range of Rs. 125 – Rs. 417 between March 2019 and May 2026. It has also issued bonus shares in the ratio of 2 for 1 in March 2016, and 9 for 1 in August 2025. The average cost of acquisition of shares by the promoters is Rs. 0.00, Rs. 0.04, and Rs. 18.34 per share.
Post-IPO, company’s current paid-up equity capital of Rs. 17.07 cr. will stand enhanced to Rs. 23.33 cr. Based on the upper band of the IPO pricing, the company is looking for a market cap of Rs. 800.16 cr.
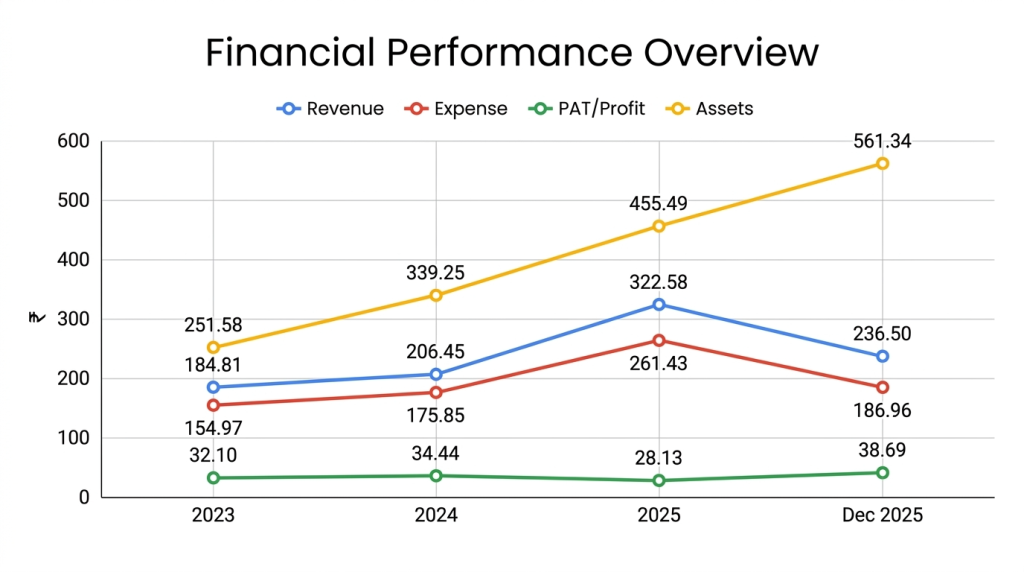
On the financial performance front, for the last three fiscals, the company has (on a consolidated basis) posted total income/ net profit, of Rs. 184.81 cr. / Rs. 32.10 cr. (FY23), Rs. 206.45 cr. / Rs. 34.44 cr. (FY24), Rs. 322.58 cr. / Rs. 28.13 cr. (FY25). For 9M of FY26 ended on December 31, 2025, it earned a net profit of Rs. 38.69 cr. on a total income of Rs. 236.50 cr. Though it posted growth in its top lines for the reported periods, its bottom line posted inconsistency. For FY25, it posted lower net profit of Rs. 28.13 cr., and for 9M-FY26, though the top line is Rs, 236.50 cr. it posted bumper profit of Rs. 38.69 cr. in a pre-IPO period, that not only raise eyebrows, but also concern over its sustainability going forward. Despite higher other income for FY25, it marked lower net following extra-ordinary item of Rs. 16.97 cr. Its contingent liability stood at Rs. 61.64 cr. as of December 31, 2025, that raises alarm. Its overall borrowings of Rs. 242.57 cr. as of December 31, 2025, raise concern.
For the last two fiscals, the company has reported an average EPS of Rs. 25.00, and an average RoNW of 23.17%. The issue is priced at a P/BV of 2.44 based on its NAV of Rs. 140.81 per share as of December 31, 2025, but its post-IPO NAV data is missing from the offer documents.
If we attribute FY26 super earnings to its post-IPO fully diluted paid-up equity capital, then the asking price is at a P/E of 15.51, and based on FY25 earnings, the P/E stands at 28.44. The issue appears fully priced, based on its bumper earnings for 9M-FY26, which may not be sustained.
For the reported periods, the company has posted PAT margins of 17.56% (FY23), 16.92% (FY24), 8.97% (FY25), 16.65% (9M-FY26), and RoCE margins of 22.14%, 19.25%, 17.66%, 13.32%, respectively, for referred periods.
All amounts in Indian Rupees crores
The company has not paid any dividends for the reported periods of the offer document. It will adopt a prudent dividend policy, based on its financial performance and future prospects.
As per the offer document, the company has no listed peers to compare with.
The two merchant bankers associated with this issue have handled 79 issues in the past three years, out of which 8 issues closed below the issue price on listing date.
QBL is engaged in the business of developing, manufacturing and marketing of diverse range of reagents and consumables. It posted growth in its top lines for the reported periods, but suffered a setback for FY25 in bottom line following accounting adjustments. As the company has no listed peers, it is trying to extract fancy price for its IPO. Based on its overall financial data, the issue appears fully priced. Well-informed investors may park moderate funds for long term.
Dilip Davda is a veteran financial journalist associated with the Indian stock market since 1978. He has been contributing to print and electronic media on capital markets, insurance, and finance since 1985.
He is widely recognized for reviewing public issues and non-convertible debentures (NCDs) in the primary market. Drawing on over three decades of market experience and close interaction with merchant bankers, his reviews focus on detailed fundamental and financial analysis of companies, with a special emphasis on SME public issues.
Disclaimer: The information provided herein is solely for educational and informational purposes and does not constitute an offer, solicitation, or recommendation to buy or sell any securities. Readers are advised to consult a qualified financial advisor before making any investment decisions. Investments in the securities market are subject to market risks. The author does not intend to invest in the securities discussed.












2 Responses
what is the buy back date of aarti drug?
g8